|
12/28/2022 0 Comments Cathode ray experiment for kids Influenced by the work of James Clerk Maxwell land the discovery of the X-ray, Thomson deduced that cathode rays (produced by Crookes tube) exhibited a single charge-to-mass ratio e m and must be composed of a single type of negatively charged particle, which he called "corpuscles." G. #CATHODE RAY EXPERIMENT FOR KIDS SERIES#Thomson's discovery of the electron began in 1895 with a series of experiments in the Cavendish Laboratory. One of his students was Ernest Rutherford, who would later succeed him in the post. In 1890, he married Rose Paget, and he had two children with her. In 1884, Thomson became Cavendish Professor of Physics. He was chosen Master of Trinity in 1918 and guided the college until shortly before his death. The highest-scoring student is named the Senior Wrangler, the second-highest is the Second Wrangler, etc.). He was accepted in 1876 and became a Fellow in 1880, when he became Second Wrangler (a student who has completed the third year of mathematics with first-class honors. His professor of mathematics recognized his brilliance, and he was encouraged to apply for a scholarship at Trinity College in Cambridge. So Thomson entered Owens College (now the Victoria University) in Manchester. His father, a bookseller, wanted him to be an engineer, but did not have the fee for J.J.'s apprenticeship. Thomson, was born on December 18, 1856, in Cheetham Hill, a suburb of Manchester, England, to Scottish parentage. If you appreciate our work, consider supporting us on ❤️ patreon.Sir Joseph John Thomson, often known as J.J. Thomson (left) with his student Rutherford Associated articles His atomic model did gain popularity until his own student Rutherford in 1911 found the presence of the nucleus through his gold foil experiment. However, the hydrogen atom can emit light at different frequencies. According to the model, the hydrogen atom can emit light only at one frequency. The model was rejected because it was ineffective to justify light spectrum.The model was abandoned as it was unable to explain the deflection of alpha particles when projected on gold foils. It failed to explain Rutherford's gold scattering experiment.We know the positive charge is centred in the atoms. The model says the positive charge is uniformly distributed throughout the atom.It did not mention the presence of the nucleus in an atom.In spite of its advantages, it has many disadvantages. Thomson's model was a milestone in the atomic world. Comparison between plum pudding and atom Limitations/drawbacks Hence, the model got its name the plum pudding model. The electrons in the cloud of uniform positive charge is similar to the plums in a pudding, a popular British dessert. There is also a repulsive force between electrons. Thus, Thomson conceptualized the atom as: …a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification…Īs per the model, the negatively charged electrons experience the net electrostatic force by the positively charged sphere towards the centre of the atom. In those days, the knowledge of the nucleus not revealed. 
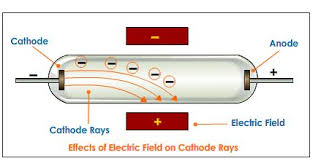
Therefore, the net charge on an atom remained zero. To explain this, he introduced the presence of the positive charge in an atom such that the positive charge counterbalances the negative charge. Using this ratio, Thomson concluded the size of these particles was even smaller than of a hydrogen ion.Īt that time, it was known the atoms were electrically neutral. He also calculated the mass-to-charge ratio of these particles. He described that cathode rays were negatively charged corpuscles, which were later renamed as electrons, and were constituents of atoms. Thomson studied magnetic and electric properties of cathode rays. Thomson's plum pudding atomic model Explanation
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
